 Here is a commonly accepted order of elements in the reactivity series: The reactivity series follows a general order, although slight variations can occur based on specific reaction conditions. This concept is essential in understanding various chemical phenomena, such as the extraction of metals, predicting reaction outcomes, and preventing corrosion. Elements at the top of the reactivity series are the most reactive, while those at the bottom are the least reactive. The series allows scientists to predict the behavior of elements when they come into contact with other substances and provides insights into the types of chemical reactions they are likely to undergo. It provides a systematic framework for comparing and ranking elements according to their reactivity. The reactivity series is a hierarchical arrangement of elements based on their relative tendency to undergo chemical reactions. After reading this article, you will be able to understand the nature of the Reactivity Series as well as its uses and functions. 
The Arabic numbering system is the most widely accepted today.In this article, you will learn about the Reactivity Series, including its significance and its applications. Many periodic tables include both Roman and Arabic numbers. The modern IUPAC system uses Arabic numbers 1-18, simply numbering the columns of the periodic table from left to right.The CAS system used letters to differentiate main group (A) and transition (B) elements.The older IUPAC system used Roman numerals together with letters to distinguish between the left (A) and right (B) side of the periodic table.Three systems have been used to number families and groups: Recognizing Families on the Periodic TableĬolumns of the periodic table typically mark groups or families. Noble Gases: - Group 18 (VIIIA) - 8 valence electrons.Halogens: - Group 17 (VIIA) - 7 valence electrons.Oxygen Group or Chalcogens: - Group 16 (VIA) - 6 valence electrons.Nitrogen Group or Pnictogens: - Group 15 (VA) - 5 valence electrons.Carbon Group or Tetrels: - Group 14 (IVA) - 4 valence electrons.Boron Group or Earth Metals: Group 13 (IIIA) - 3 valence electrons.Transition Metals: Groups 3-12 - d and f block metals have 2 valence electrons.Alkaline Earth Metals: Group 2 (IIA) - 2 valence electrons.Alkali Metals: Group 1 (IA) - 1 valence electron.Many chemists and chemistry textbooks recognize five main families:Īnother common method of categorization recognizes nine element families: 
However, there are different ways of categorizing elements into families. Because element properties are largely determined by the behavior of valence electrons, families and groups may be the same. 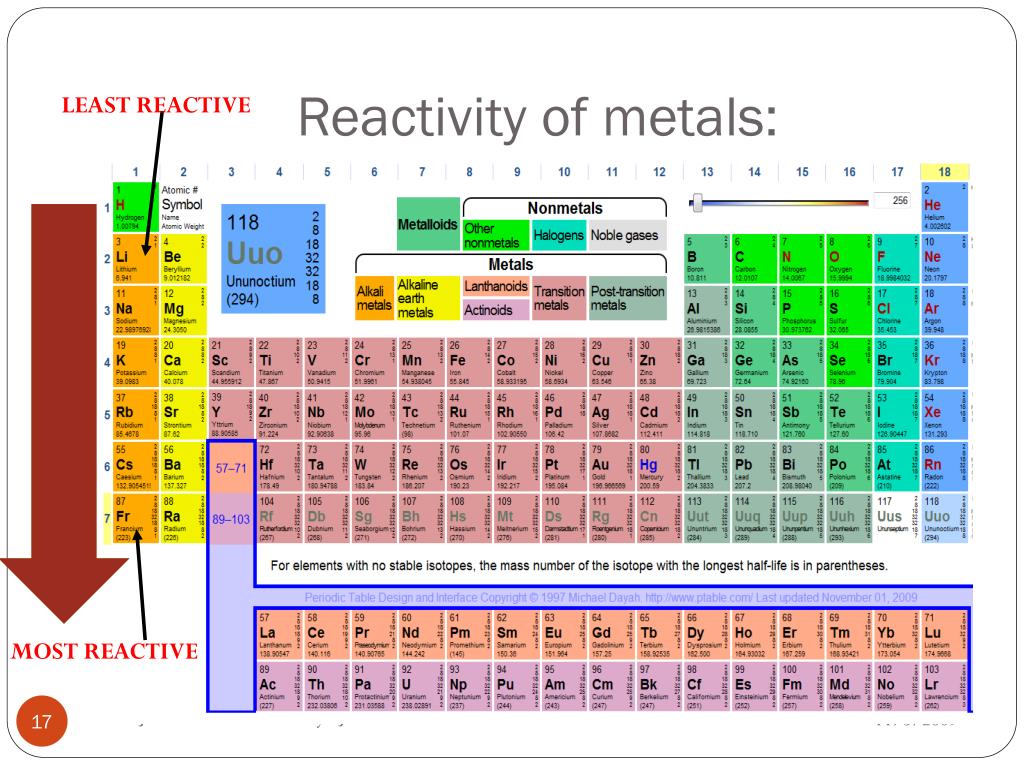
Element groups, on the other hand, are collections of elements categorized according to similar properties. 
The characteristics of the elements in these families are determined primarily by the number of electrons in the outer energy shell. Elements are classified into families because the three main categories of elements (metals, nonmetals, and semimetals) are very broad. Element families are indicated by numbers located at the top of the periodic table.Īn element family is a set of elements sharing common properties. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
